Novartis will make case to FDA for approving pill-based multiple sclerosis treatment
By APTuesday, June 8, 2010
SUMMARY BOX: Novartis’ multiple sclerosis drug
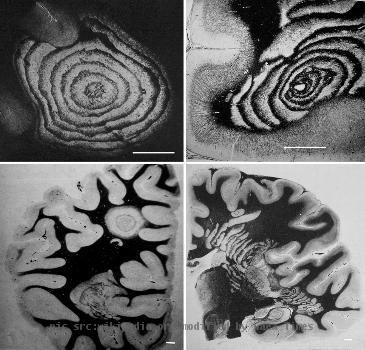
Novartis is moving closer to marketing a pill-based drug for multiple sclerosis, though a federal review released Tuesday underscored safety concerns with the medication.
NEW OPTION: Currently all the drugs used to treat the disease are injectables. Novartis’ Gilenia would be the first pill for the disease.
SAFETY CONCERNS: FDA staff noted side effects with the drug, including eye disorders, heart problems and weakened lung function.
EXPERT OPINION: On Thursday the FDA will ask a panel of outside advisers to vote on whether Gilenia should be approved. The FDA is not required to follow the group’s advice, though it usually does.
Filed under: FDA, Multiple Sclerosis
Tags: Diagnosis And Treatment, Diseases And Conditions, Medication, Multiple Sclerosis
Tags: Diagnosis And Treatment, Diseases And Conditions, Medication, Multiple Sclerosis
YOUR VIEW POINT